Training Guideline FDA 21 CFR Part 11, eLearning
600033-0201-904
- product type
- eLearning
- Training type
- eLearning
- Level
- 1
Overview about guideline 21 CFR Part 11
Code of Federal Regulation of Food and Drug Administration (FDA)
- description of several approaches to reduce measurements errors
- correct interpretation of single section in 21 CFR Part 11.
- specific description of requirements of 21 CFR Part 11 when applied to a process or application
Duration: approx. 1 hour
Product note
ATTENTION! Contents of this eLearning only available in English language.
Recently Viewed
Training Guideline FDA 21 CFR Part 11, eLearning
600033-0201-904
₨ 13,260.00GST Extra as appliacable
Loading (MAN) - ShuttleLoad (module 3a)
602701-9021-405
On Request
Inquire on Delivery
Live Chat Now
Cone receiver, M5 system
626107-1015-105
₨ 6,830.00GST Extra as appliacable
Inquire on Delivery
Live Chat Now
Plenum-rated sensor bus cable (10 meters)
626140-9149-101
₨ 19,090.00GST Extra as appliacable
Inquire on Delivery
Live Chat Now
End angle piece with cone adapter, M5
626107-6280-010
₨ 4,940.00GST Extra as appliacable
Inquire on Delivery
Live Chat Now
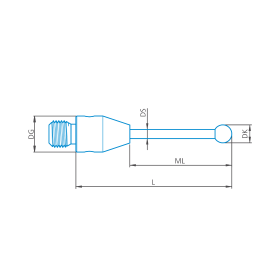
Stylus straight M3, DK2 L22
602030-8104-000
₨ 2,530.00GST Extra as appliacable
Inquire on Delivery
Live Chat Now