Training Guideline FDA 21 CFR Part 11, eLearning
600033-0201-904
- product type
- eLearning
- Training type
- eLearning
- Level
- 1
Overview about guideline 21 CFR Part 11
Code of Federal Regulation of Food and Drug Administration (FDA)
- description of several approaches to reduce measurements errors
- correct interpretation of single section in 21 CFR Part 11.
- specific description of requirements of 21 CFR Part 11 when applied to a process or application
Duration: approx. 1 hour
Product note
ATTENTION! Contents of this eLearning only available in English language.
Recently Viewed
Training Guideline FDA 21 CFR Part 11, eLearning
600033-0201-904
₨ 13,260.00GST Extra as appliacable
DAkkS calibration, Step Guage Level 0, 700mm
600080-0010-014
₨ 125,490.00GST Extra as appliacable
Inquire on Delivery
Live Chat Now₨ 162,790.00GST Extra as appliacable
Inquire on Delivery
Live Chat Now
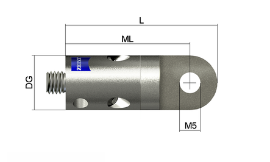
End angle piece, rotating , M5
626105-6280-010
₨ 10,300.00GST Extra as appliacable
Available
Quick change unit V3, upper part, 80 mm
600062-0205-016
₨ 13,130.00GST Extra as appliacable
Inquire on Delivery
Live Chat Now
THETA 55 pallet, M6 50x50, for MICURA
626109-9210-050
₨ 102,970.00GST Extra as appliacable
Inquire on Delivery
Live Chat Now
THETA 55 pallet, 1/4-20 and 1/2x1/2 grid, Aluminum
626109-9220-530
₨ 160,250.00GST Extra as appliacable
Inquire on Delivery
Live Chat Now
OmniFix Three-jaw ring chuck mini Ø30 mm
626109-9220-082
₨ 114,550.00GST Extra as appliacable
Available
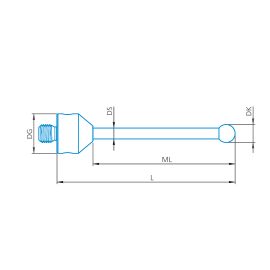
Stylus straight M5, DK5 L75
602030-9015-000
₨ 4,280.00GST Extra as appliacable
Available